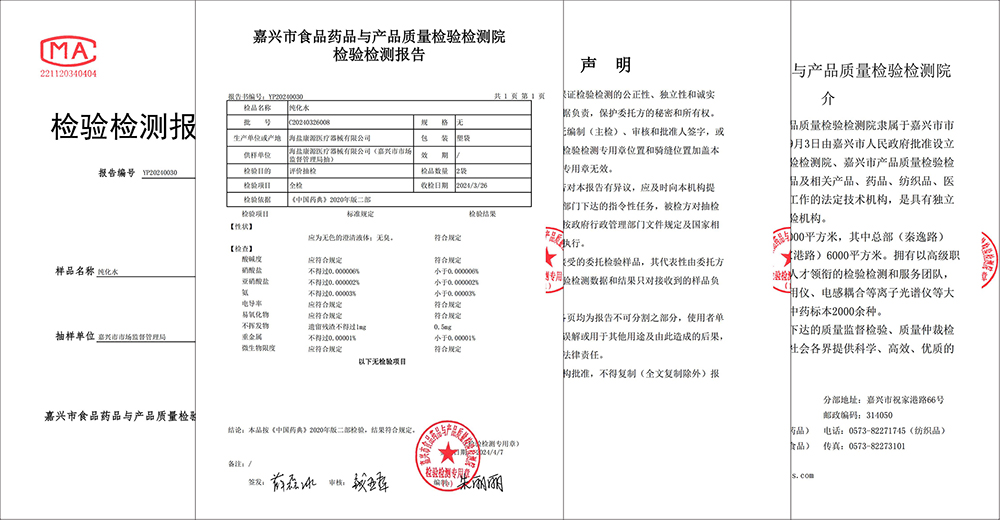
Viru kuerzem huet d'Jiaxing Market Supervision Administration eng ëmfaassend Proufnahme vum Prozesswaasser vun der Haiyan Kangyuan Medical Instrument Co., LTD. duerchgefouert an ugekënnegt, datt d'Prozesswaasser vu Kangyuan Medical vollstänneg mat den Ufuerderunge fir gereinegt Waasser vun der Editioun 2020 vun der chinesescher Pharmakopöe entsprécht, wat déi exzellent Kontrollfäegkeet vu Kangyuan Medical a punkto Produktqualitéit a Patientesécherheet validéiert.
D'Proufkontroll gouf vun der Jiaxing Market Supervision Administration organiséiert an vum Jiaxing Food, Drug and Product Quality Inspection and Testing Institute an Optrag ginn. Am Aklang mat de jeeweilegen nationalen Normen an Industrienormen huet den Inspektiouns- an Testinstitut en ëmfaassenden an professionellen Test vum Prozesswaasser duerchgefouert, dat vu Kangyuan Medical fir d'Produktioun vu verschiddene medizineschen Apparater benotzt gëtt, dorënner de pH-Wäert vum Waasser, Nitrat, Konduktivitéit, Schwéiermetaller, mikrobiell Grenzen a vill aner Aspekter. No e puer Ronne vu rigoréisen Tester weisen d'Resultater, datt d'Prozesswaasser vu Kangyuan Medical d'Ufuerderunge fir gereinegt Waasser vun der Editioun 2020 vun der chinesescher Pharmakopöe vollstänneg erfëllt, wat d'Qualitéit, d'Sécherheet an d'Zouverlässegkeet vu Kangyuan medizineschen Apparater vollstänneg garantéiert.
Kangyuan Medical huet d'Produktqualitéit an d'Patientesécherheet ëmmer un den éischte Plaz gestallt a leet besonnesch Wäert op d'Qualitéitskontroll vum Prozesswaasser. D'Firma huet fortgeschratt Waasserproduktiounsausrüstung an Iwwerwaachungstechnologie agefouert, an e solides Prozesswaassermanagementsystem a Betribsprozeduren etabléiert fir sécherzestellen, datt all Drëps Waasser den nationale Standarden entsprécht. D'Besteetung vun der Proufnahmeinspektioun ass net nëmmen eng Bestätegung vun der Qualitéit vum Kangyuan Medical Prozesswaasser, mä och eng Unerkennung vum allgemenge Qualitéitsmanagementsystem vu Kangyuan Medical.
An Zukunft wäert Kangyuan Medical seng déifgräifend Industrieakkumulatioun an de Geescht vun der kontinuéierlecher Innovatioun bäibehalen, weiderhin eng féierend Roll an der Medizinindustrie spillen, fir sécherzestellen, datt d'Majoritéit vun de Patienten iwwer besser a sécher medizinesch Verbrauchsmaterialien hëllefe kënnen, a méi e grousse Bäitrag zur mënschlecher Gesondheet leeschten.
Zäitpunkt vun der Verëffentlechung: 29. Mee 2024

 中文
中文